THESE BUBBLES KILL CANCER
It’s called histotripsy. And it could change cancer treatment forever.
By the time Carrie Kumpel and her husband, Albert, touched down at Detroit Metro Airport in May 2022, her body had already endured more than five years of the push and pull, short highs and long lows of sustained cancer treatment.
Contents:
Various tumors had come and gone and come again over that stretch—in her bones, her lymph system and, persistently, in her liver. Each brought its own treatment regimen—hormones, chemotherapy drugs, radioactive pills—with different side effects and corresponding shifts in her quality of life.
Despite it all, the Bakersfield, California resident was a stage IV cancer patient—one who had already lost so much. The high school English teacher’s natural hair had gone, along with her ability to bear children.
“You learn to do your living between scans,” said Carrie, 40. “But here I am, six years after diagnosis, which is pretty lucky.”
On this day, the couple headed west from the airport to Ann Arbor and the University of Michigan’s Rogel Cancer Center. Carrie had agreed to participate in human trials for a new treatment called “histotripsy”— a technique created by U-M engineers and doctors that harnesses soundwaves to attack cancer. It comes with the promise of few, if any side effects, a quick treatment time and, for Carrie, the hope that it would completely destroy three spots that had formed on her liver.
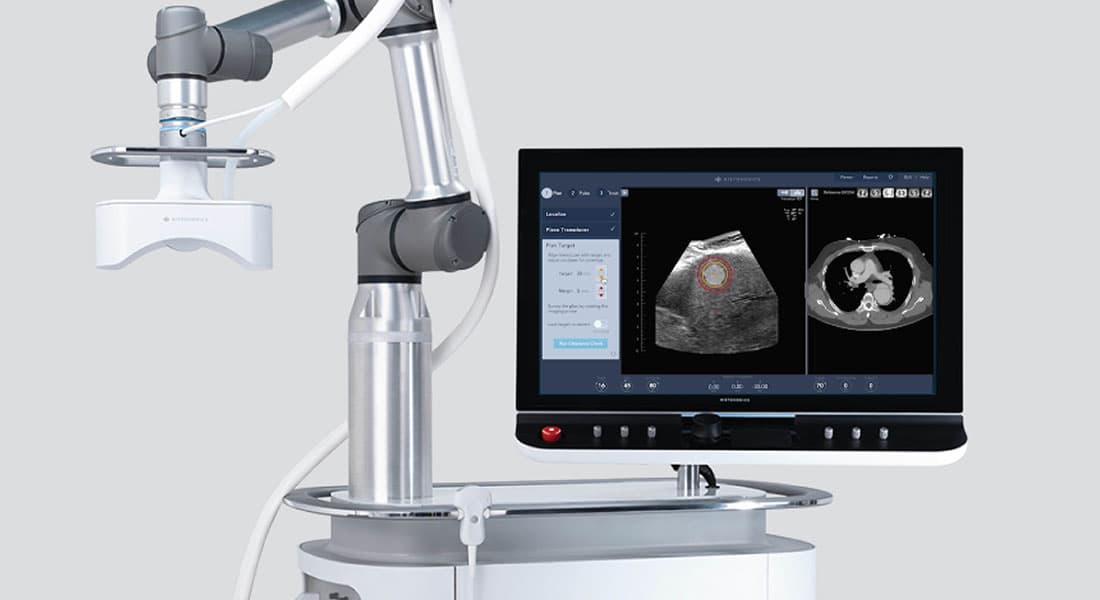
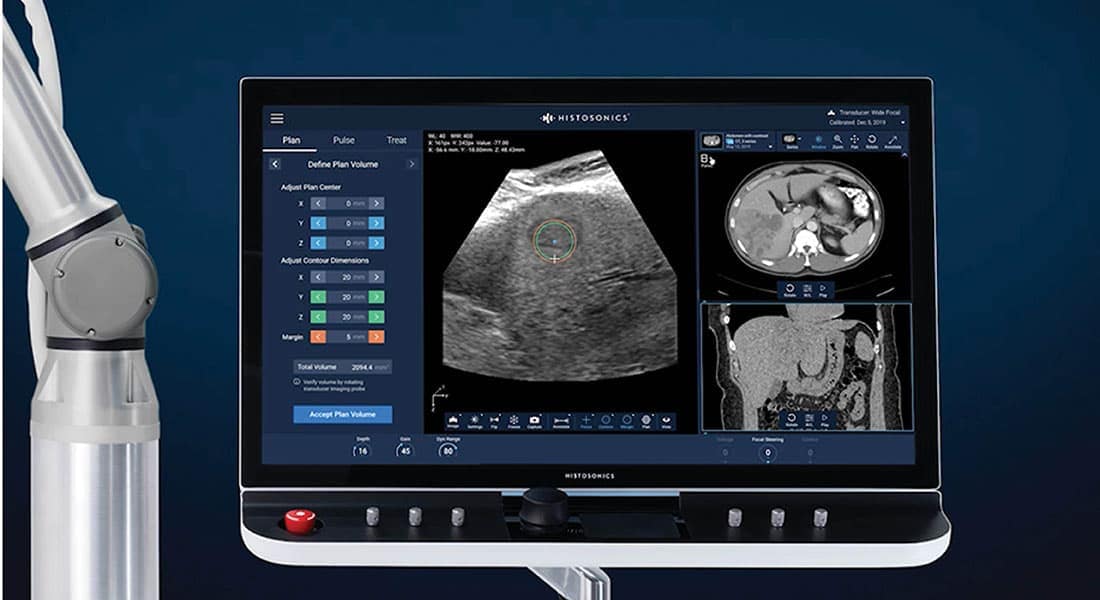
Her doctors put her under anesthesia for the procedure in order to keep her body still and her breathing normal. While Carrie was asleep, the medical team began by moving an ultrasound probe across her abdomen, over the liver tumor within, to identify the precise location of the targets.
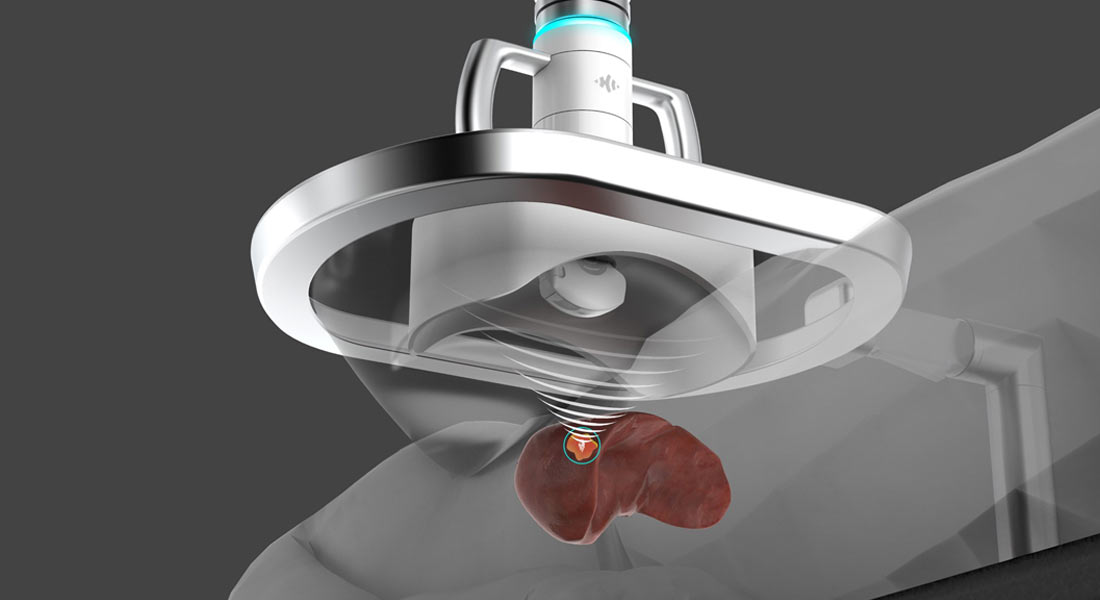
As she continued to sleep, a larger ultrasound transducer attached to a robotic arm took its turn hovering over the targeted spot on Carrie’s abdomen, delivering sound waves toward the cancerous lesion below. No incisions, no medicines, no scar tissue and no radiation.
When she woke up a short time later, there was nothing to indicate that any treatment had taken place.
“I really didn’t have any residual sickness or pain,” she recalled in July 2023. “I felt fantastic the next day. A day or two after the procedure, I was able to go visit the Henry Ford Museum.”
One month later, an MRI showed what Carrie and Albert had hoped for: The targeted mass had been eliminated by histotripsy. One simple procedure in one afternoon.

Her treatment was part of human trials in the United States and Europe which, in recent years, have helped bolster histotripsy’s case with regulators at the U.S. Food and Drug Administration (FDA). The results show histotripsy delivering on its promise—destroying tumors with few, if any, side effects.
A private company, HistoSonics, was co-founded in 2009 by U-M engineers and doctors to commercialize the technology. On the strength of the initial human trial results, the company received formal approval from the FDA in October to market histotripsy for liver treatments in humans.
THE SCIENCE IS
SOUND
The procedure worked as planned, striking and reducing the tumor 95.5% of the time.
It’s a major step toward bringing the technology into the mainstream. By using soundwaves—the same technology we trust to give us our first peek at new life in the womb—Carrie and other patients could be spared the physical and mental toll that comes with traditional treatments like surgery, chemotherapy and radiation.
Cancer’s deadly toll
Worldwide, roughly 10 million people die from cancer each year. In the United States alone, an estimated two million will be diagnosed with cancer in 2023. Another 609,000 Americans are expected to die from it, keeping cancer the second leading cause of death behind heart failure.
The late Charles Cain (PhD EE ’72), the “father” of histotripsy and a long-time U-M professor of biomedical engineering and electrical engineering and computer science, saw it as “a knife-less surgical approach” that leaves behind “no recognizable tissue fragment.” Cain guided the research at U-M from its humble beginnings to the brink of FDA approval before his passing three years ago.
Much more than knifeless, histotripsy comes with no damage to surrounding tissues, none of the side effects of radiation and surgery and little to no recovery time. It’s the kind of procedure researchers feel can eventually be conducted in a medical office instead of an operating room. As HistoSonics CEO Mike Blue told the BBC earlier this year, “Patients will awake from their procedure and, generally, most times, not know that they were treated.”
Both the inventors of the treatment and the company bringing it to the world have unabashedly high hopes for what histotripsy can become.
“In the long run, I envision histotripsy providing incisionless, non-toxic, painless and low-cost treatment to cancer patients at major hospitals as well as local clinical centers across the world,” said Zhen Xu, a U-M professor of biomedical engineering and an inventor of the histotripsy treatment. “I also hope to see histotripsy used to boost immunotherapy to treat tumor metastases and be extended to treat patients with many other disease types beyond cancer, such as stroke, neurological diseases, cardiovascular diseases and skin diseases.”
Despite gains in reducing deaths, cancer continues to throw curveballs. Late in 2022, an analysis of data from 44 countries showed that diagnoses for more than a dozen types of cancer were on the rise in people under the age of 50.
But histotripsy doesn’t need to understand cancer to be effective. Its reliance on physical force generated by sound means it can ignore questions like what turns normal cells malignant or why certain abnormal cells grow and spread in the body. It’s simply good at killing a killer.
Carrie Kumpel remains a stage IV cancer patient. The questions surrounding her current and future treatments will have more to do with maintaining her condition than curing it.
“If more people had access to this kind of treatment, something that comes with so much less inflammation and damage, I think it would be really amazing,” she said.

Turning bubbles into weapons
When you’re a toddler, bubbles have a magic quality about them. Creating clouds of them with bubble soap wands can fascinate for a whole afternoon. And popping them? Even better.
But when you focus them on a cancerous tumor in just the right way, they can become a deadly weapon.
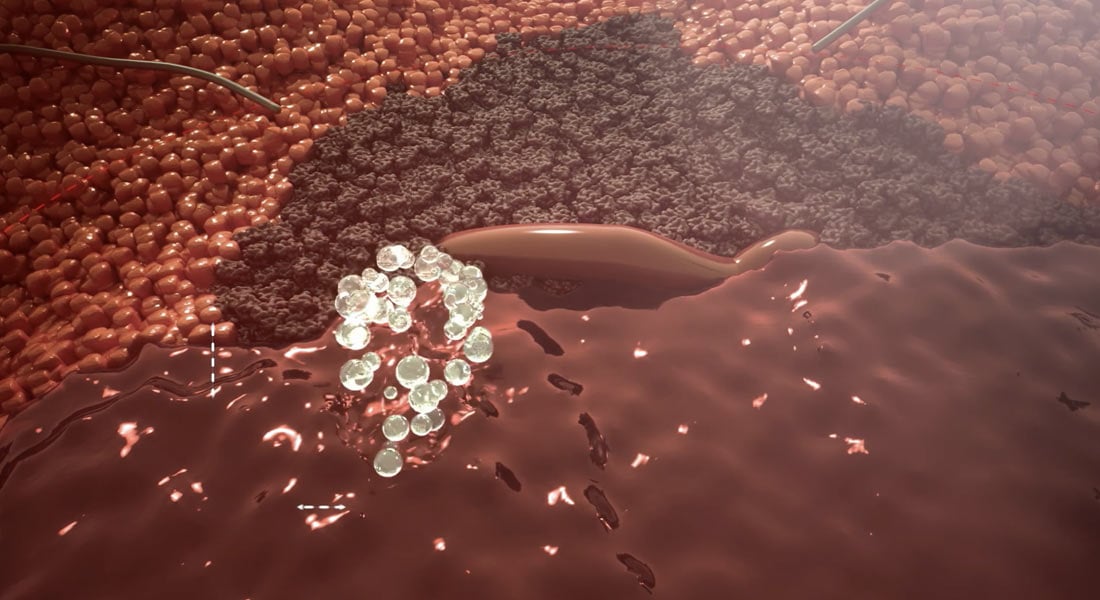
The process uses a transducer—which converts electricity into sound—to deliver ultrasound waves to a malignant mass at a precise location. When the waves hit gasses inside cancerous cells, they generate clouds of tiny bubbles through a process known as cavitation.

Pulsing sound waves causes the millimeter-sized bubble clouds to repeatedly grow and collapse. On an ultrasound monitor, it can look like bubbles from boiling water—quickly rising and falling along the surface in your pot.
In the past, researchers saw the creation of bubbles through ultrasound as “uncontrollable,” something to be avoided. Histotripsy, however, generates mechanical energy to activate those bubble clouds and break up the tumor cells’ structure, turning it into a liquid called acellular lysate.
“It’s just fluid—the denatured proteins that the body recognizes as foreign material,” said Dr. Mishal Mendiratta-Lala, a U-M professor of radiology who performed Kumpel’s histotripsy procedure. “And unlike other treatments, histotripsy does not destroy all nearby blood vessels. This preserves blood flow in the treatment zone to carry acellular lysate away—essentially removing the dead tumor in the form of this fluid—and the whole area goes back to normal.”

Histotripsy also uses ultrasound to give physicians a detailed look at the cancerous mass before and during treatment. This enables them to map out treatment before it starts and monitor the process while it’s happening. The result is startling accuracy.
The cellular and subcellular destruction caused by histotripsy is limited to the target area. In fact, the target diameter of a histotripsy pulse can be no more than one millimeter. It’s that kind of precision that helps set histotripsy apart from traditional cancer treatments like chemotherapy and radiation—which employ more of a carpet-bomb approach that can damage areas outside the intended tumor.
Human trials of histotripsy treatments for cancer began in 2020 and 2021, when doctors in Europe and the United States began a trial called #HOPE4LIVER. With COVID-19 making every plan uncertain, HistoSonics worked with multiple hospitals and medical schools on both sides of the Atlantic to locate and treat patients. Researchers zeroed in on liver cancer to provide a strong test of the technology.
Liver cancer is particularly tough to treat successfully—the five-year survival rate is less than 20%. Only about 20% of liver cancer patients are eligible for surgery, meaning the majority of those patients don’t have a surgical option.
Locations in Europe and the United States treated 44 different patients with a total of 49 lesions. Several of those patients received their histotripsy at Michigan Medicine. Doctors were able to successfully target tumors in 42 of the 44 patients, with the two “misses’’ attributed to physician error. And the procedure worked as planned, striking and reducing the tumor 95.5% of the time.
Another indicator of human trial success is how many adverse events of any level of severity—from a high temperature to a toothache—crop up among the patient pool in the month following treatment. In the #HOPE4LIVER Trial, procedure related complications requiring medical intervention were reported in only 3 of 44 study participants.

Outsmarting cancer
Cancer can be sneaky and elusive. Among its most insidious qualities, a trait that drastically hinders the body’s chances of fighting it, is its ability to hide.
Look inside a cancer cell and you’ll find tumor antigens—proteins specific to cancer. When the body’s immune system detects tumor antigens, it responds by attacking the cells. But the body often can’t see through a cancer cell’s outer wall to identify tumor antigens. Star Trek fans might think of those walls as a “cloaking device”—granting cancer the invisibility it needs to grow unchecked without alerting the body’s immune system.
It’s one of the reasons many cancers aren’t detected until after they’ve spread to problem areas in the body and put the host’s life at risk. But histotripsy foils cancer’s cloaking efforts by destroying its cell walls, leaving the tumor antigens in plain sight for the body’s immune system.
This effect was detailed in a pair of papers published by the U-M research team between March 2022 and January 2023. They demonstrate that the sound waves used to break down cancerous tumors in rats also helped trigger the rats’ immune response. After histotripsy destroyed 50% to 75% of liver tumor volume, the rats’ immune systems cleared away the rest, with no evidence of recurrence or metastases in more than 80% of animals.
That immune response occurred throughout the body, not just in areas targeted by the histotripsy treatment, resulting in the reduction of tumors far from the treated area.
With human trials utilizing histotripsy only getting underway in recent years, the data in this area of research is still limited. But it raises the tantalizing possibility that the treatment could be transplantable, according to recent testing in mice.
After breaking up a tumor in one mouse using histotripsy, the team extracted some of the tumor lysate and injected it into another mouse. Both mice developed immune protection from that cancer.
“Injecting the tumor lysate into a second mouse had almost a vaccine-like property,” Xu said. “Mice that received this debris were surprisingly resistant to the growth of cancers.”
A better way to treat
Carrie Kumpel can rattle off the medications and treatments she’s had over the years—most with bizarre, vowel-intensive names—as if she’s naming family members.
And while she hasn’t always had to endure their worst side effects, none have created the kind of experience she’d want to repeat. But endurance is key for cancer patients because, all too often, traditional treatments can continue for years.
For her early breast cancer treatments there were drugs like Xeloda and Cyclophosphamide, which Carrie described as “pretty awful.” Treatments like Zoledronic acid injections are designed to prevent the spread of breast cancer, but can also cause a host of side effects that patients struggle to deal with.
Then there’s Doxil, which some patients refer to as the “Red Devil,” for its red coloring as well its propensity for causing red urine, hair loss, mouth sores, nausea and vomiting.
Another “favorite” from Carrie’s past is Y90, a liver-specific treatment delivering radioactive beads into blood vessels near malignant lesions. Patients are usually instructed to avoid strenuous activity for ten days following insertion, and it can take up to three weeks to return to normal function.
“I’ve been in constant treatment for five-plus years—even when I don’t have new cancer popping up,” she said. “Even when I don’t have anything active, it’s required for me to still be in some kind of chemo because that’s the way they do it.”
I envision histotripsy providing incisionless, non-toxic, painless and low-cost treatment to cancer patients.
According to the American Cancer Society, cancer patients deal with a wide range of side effects depending on the treatment.

Chemotherapy can cause chemo brain (impacting concentration and focus), nausea and vomiting, nerve problems, fertility issues, hair loss, bladder issues, kidney problems, changes in libido and sexual function and others.
Radiation directed at different parts of the body can bring different impacts. The scope of side effects include: nausea, earaches, tooth decay, hair loss, loss of appetite, bladder problems and fertility problems.
Surgery’s impacts include pain, extended recovery times, swelling, drainage, fatigue, appetite loss, infection and others.
These traditional treatment options, along with their physical and mental costs, are what histotripsy seeks to replace. The pending FDA approval would clear the way for liver treatments specifically.
With continued success, HistoSonics officials hope the technology will also be approved for other treatments, from brain tumors to heart blockages.
The breakthrough
In the span of a few hours over one afternoon in the lab on North Campus, weeks spent scouring slaughterhouses for pig parts suddenly seemed to pay off. At least that’s what Xu hoped as she walked, porcine heart in hand, down the short corridor from the lab to Charles Cain’s office. The heart had three fresh holes in it, the result of what might be considered the first successful test of histotripsy in the lab.
This was in late April of 2002. Xu had spent weeks working in the North Campus lab, trying out new combinations of amplitudes and frequencies to elicit the destruction of small sections of the pig heart, which was standing in for human tissue.
Xu had become all too familiar with the art of acquiring animal organs for these experiments. The team’s early work was focused on congenital heart disease so, each week, she would visit a slaughterhouse 15 miles south in Milan, Michigan in search of pig hearts to test.
“The first time there, the man showed me a trash can filled with pig parts and said ‘See what you want, go ahead and get it,’” Xu recalled. “It didn’t smell so good since there were intestines and other things in the can. It was quite bloody. Now, in some cases, they would have cut through the hearts, so I’d search through them looking for ones that weren’t quite as damaged. Back then, we could get two, maybe three pig hearts for $5.”
For Xu, there was nothing off-putting about this part of her job; she grew up in Nanjing, China and her father worked on a farm for a time. She would cut the hearts into smaller sections and fix one of those sections inside a water tank as the experiment’s stand-in for heart tissue.
Water played the role of healthy tissue surrounding the heart, since ultrasound waves won’t propagate through the air. Next, Xu lined up an ultrasound transducer, which turns electricity into ultrasound waves.
Xu had little experience with ultrasound when she hired on with Cain’s team as a PhD student research assistant. In fact, her lack of experience was a selling point for the man who would become her mentor. He wanted someone who wasn’t constrained by previous research in any way.
As a result, that day in 2001, Xu decided to try something new—hooking her transducer up to an amplifier that no one else seemed to be using because it was too loud. A lab partner quickly reaffirmed that the machine was, indeed, too loud. So Xu began operating it in the 20 kilohertz pulse repetition range, right above the range of human hearing, with a camera capturing the action.
She fired micropulses of soundwaves—super-short bursts—at amplitudes much higher than normal. She figured those bursts would make any noises less likely to draw the ire of coworkers.
After dozens of previous attempts with different parameters, Xu wasn’t expecting fireworks. But after a short time, she noticed a hole had appeared in the pig heart.
“My initial reaction was ‘Is that real?’” she recalled. “Then my mind immediately shifted to ‘Can I repeat that? If I can’t repeat it, it’s not real.’”
Two more successful tries, and Xu was off down the hallway with her prized pig heart section to share what happened.
“We found that ultrasound somehow generated those holes,” she said. “Then we spent the next ten years trying to discover how it worked.”
The “father” of histotripsy
Everyone seemed to take something different away from their time with Charles Cain.
In the early aughts, students posting anonymously on the Rate My Professors website noted that Charles Cain “looks like Santa Claus,” and that he “teaches from notes that are so old, they are yellow.”

Michigan Engineering leaders focused on more relevant aspects like Cain’s leadership. In 1996, they tapped him to be the first chair of the newly-created Department of Biomedical Engineering.
Today, fellow researchers speak of his focus—a tunnel vision in his work that was both admirable and productive, but also capable of coming across as gruff and uncompromising.
Tim Hall’s office displays his fondness for Cain through a handful of Easter eggs—a nameplate on a shelf and a copy of Cain’s PhD dissertation nearby. One of the founders of HistoSonics, Hall (BSE ECE ’98, MSE ’01, PhD BME ’07) is a U-M research scientist in biomedical engineering who worked with Cain for decades.
“Some of the adjectives I’d use to describe him are ‘maverick,’ I think that’s a good term,” Hall said “He was definitely a fierce advocate.” Then, jokingly: “He loved to argue and discuss ideas. Or, at least, his ideas.”
Born in Tampa, Florida and raised in Miami, Cain earned his bachelor’s degree from the University of Florida, his master’s from MIT and his PhD from U-M, all in electrical engineering. He titled his 1972 PhD thesis “A Control Systems Study of the Human Oculomotor Predictive Tracking Mechanism.”
Before earning his PhD, Cain worked at Bell Laboratories in Naperville, Illinois, focusing on solid-state electronic switching systems. From 1972 to 1989, he was a professor of electrical and biomedical engineering at the University of Illinois at Urbana-Champaign.
There he worked with Bill and Frank Fry, brothers who helped pioneer the use of ultrasound in medical diagnostics and surgery, with a particular focus on the brain. Cain brought his expertise in ultrasound technologies to U-M in 1989.
He had a tender, tender heart that I think only those closest to him saw.
When Hall began working with the team in the late 1990s, Cain had been focused on treating tissues in the heart with the heat generated by ultrasound.
“By 2000, Charles had mostly given up on that technology,” Hall said. “He had some students who had used modeling to show that it wasn’t a practical approach for many reasons. So, he latched on to the idea that we could use cavitation to break some of these barriers that the physics of thermal ablation were imposing on it.
“The effects of cavitation had been noted back in the 1960s and 1970s—that they could accompany thermalization when the ultrasound power was very high. But at the time, cavitation was a little bit unpredictable.”
The quest to find the right way to control cavitation would prove difficult. But Cain’s focus and drive remained constant. As Xu wrote in memoriam: “Charles was born to be a scientist and educator. He only wanted to work on new concepts and despised being a follower … [He] was an excellent and caring mentor to his students and junior faculty, many of whom have become leaders in their fields. Charles was a visionary, a fearless leader and a force to be reckoned with.”
Where Cain’s research focus and drive came across as off-putting to some, Li Wu saw something different.
“He had a tender, tender heart that I think only those closest to him saw,” said Wu, who met Cain in 2006 when she was working at Siemens in Ann Arbor. She held a master’s degree in computer science which led to deep conversations—talks that gave her a peek behind Cain’s outer persona.
Their friendship evolved into more, with a wedding in 2008 followed by a decade and a half together. In the last three of those years, the couple dealt with Cain’s progressing prostate cancer. He passed away March 27, 2020 at the age of 77, survived by Wu, three sons from a previous marriage and two grandchildren. But he lived to see histotripsy begin to have the practical impact he’d always hoped for.
“He would be very, very happy to see the technology now being used on helping patients,” Wu said. “That was truly always his goal. He wanted to do research that had some impact for people in the world.”
The company
Dinner and drinks at the Carlyle Grill off Jackson Road on Ann Arbor’s west side. That’s how the founding team of U-M engineers and doctors celebrated the creation of HistoSonics in 2009.
It was a small moment of victory, reached with early support from the U-M Coulter Translational Research Partnership Program, which specializes in helping researchers develop and commercialize healthcare products. The program connected the histotripsy team with early consulting help and followed up with $300,000 in funding for early testing.
Later in the company’s timeline, U-M’s Innovation Partnerships provided intellectual property support.
Since those earliest days, HistoSonics has set up its corporate headquarters in Minneapolis and bolstered its leadership with board members that represent healthcare heavyweights like Johnson & Johnson, Varian Medical Systems (now part of Siemens) and Lumira Ventures.
Yet the connection with U-M and Ann Arbor remains strong. The company founders remain shareholders and, in some cases, continue to serve as consultants. Most of HistoSonic’s research and development work takes place in offices and labs on Liberty Avenue, not far from where the celebratory dinner took place.
The company recently entered into a contract to renovate and inhabit new office and research space on Oak Valley Drive on the city’s south side. The 25,000-square foot facility is expected to open in 2024 with 50 employees, and the company plans to eventually employ 70 at the site.
Jon Cannata is HistoSonics’ director of research and advanced development. He has his own U-M connection through his wife, Laura Cannata (BA Psychology, BA Spanish ’98).
“Even to this day, the way we’ve set ourselves up being very close to the University of Michigan and our team here, it’s very synergistic,” Cannata said. “They’re working on things that are advancing the science, doing their own research and development to develop the technology that could become our next product.”
I’m not sure I ever thought I’d have the opportunity to work on something that’s truly a medical frontier.
Thai-Son Nguyen (BSE BME ’16, MS ’18) is a member of that team—a man doing a job he feels he’s been uniquely prepared for in his 34 years. Since his earliest days, he has battled a variety of health issues, requiring multiple surgeries and a drastically restricted diet.
Nguyen entered Michigan Engineering in 2007 as an undergraduate, but didn’t earn his bachelor’s degree until 2016 due to pauses in his studies caused by his medical conditions. A master’s degree in biomedical engineering followed two years later.
“Even now as an adult, I have issues,” he said. “But it was because of all of the work that has gone into science and medicine that my doctors were able to diagnose those issues and come up with the treatments and lifestyle adjustments that have allowed me to be where I am now.”
Nguyen would like his work to do something equally positive for other people. He is one of roughly a dozen U-M alumni or students currently working at HistoSonics. And, for him, it’s the best place to be at the best possible time.
“What we’re doing here is great,” Nguyen said. “During college, I’m not sure I ever thought I’d have the opportunity to work on something that’s truly a medical frontier—something so novel like this.”
Editor’s note: A previous version of this story incorrectly summarized results in the #HOPE4LIVER Trial.